FCO 9XXX-0002, Build up of residue resulting in funct failures
Order Number: EK-KA901-MG
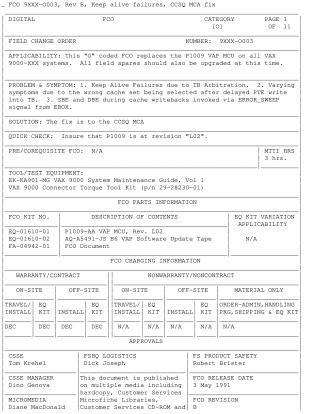
This Field Change Order (FCO 9XXX-0002), released by Digital on April 1, 1991, addresses a critical manufacturing defect affecting Microcontroller Units (MCUs) in VAX 9000 systems. The problem stemmed from uncleaned pedestals during the encapsulation process, which led to residue buildup that caused the die to come into contact with the syringe needle point, potentially resulting in gross functional failures and long-term reliability issues.
The FCO mandates upgrading all systems and field spares containing these suspect MCUs, as identified by serial numbers listed in Appendix A. The solution involves replacing the faulty Logic MCUs. The document provides a detailed procedure for MCU removal, replacement, and post-installation diagnostics, including running specific commands, MACRO level diagnostics, and a 6-hour User Environment Test Program (UETP). Field engineers are instructed to report this activity using a LARS form. Appendix A lists 69 affected MCUs found in customer systems, known upgrade sets, and those shipped as spares or upgrades with unknown locations across various regions including USA, GIA, and Europe.
Site structure and layout ©2025 Majenko Technologies